 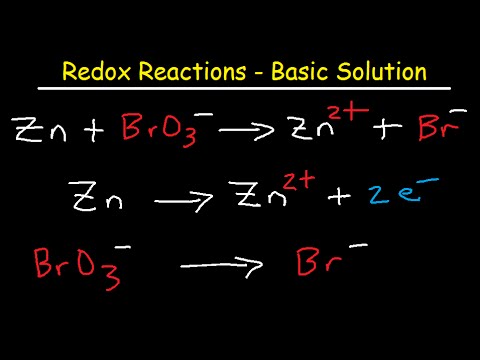 
This is not the case for the aluminum and silver reaction: the Al atom loses three electrons to become the Al 3+ ion, while the Ag + ion gains only one electron to become elemental silver. Something is amiss with this chemical equation despite the equal number of atoms on each side, it is not balanced.Ī fundamental point about redox reactions that has not arisen previously is that the total number of electrons being lost must equal the total number of electrons being gained for a redox reaction to be balanced. However, if you look at the total charge on each side, there is a charge imbalance: the reactant side has a total charge of 1+, while the product side has a total charge of 3+. Consider this redox reaction: Al + Ag + → Al 3+ + AgĪt first glance, this equation seems balanced: there is one Ag atom on both sides and one Al atom on both sides. Some redox reactions are not easily balanced by inspection. The first thing you should do when encountering an unbalanced redox reaction is to try to balance it by inspection. This redox reaction is now balanced.īalance this redox reaction by inspection. This gives us two S atoms on both sides and a total of six O atoms on both sides of the chemical equation. We can balance both the elements by adding coefficient 2 on the SO 2 on the reactant side: 2SO 2 + O 2 → 2SO 3 This now gives us six O atoms on the product side, and it also imbalances the S atoms. Clearly we need more O atoms on the product side, so let us start by including the coefficient 2 on the SO 3: SO 2 + O 2 → 2SO 3 However, the reactant side has four O atoms while the product side has three. There is one S atom on both sides of the equation, so the sulfur is balanced. Step 3.Balance this redox reaction by inspection. This gives us total changes of +2 and -2. You need 2 atoms of #"H"# for every 1 atom of #"Zn"#. Equalize the changes in oxidation numberĮach #"Zn"# atom has lost two electrons, and each #"H"# atom has gained one electron. Identify the atoms that change oxidation number Here's how the oxidation number method works for a very simple equation that you could probably balance in your head. The general idea is that electrons are transferred between charged atoms. 
The oxidation number method is a way of keeping track of electrons when balancing redox equations. None of it will happen if you don't get the oxidation number of every player in the reaction. It's just one process and one method with variations. Sometimes one method is more convenient than the other method. In both methods, you must know what the oxidation numbers are and what they become during the reaction. Then you add the two half reactions together and balance the rest of the atoms. Then you multiply them by small whole numbers to make the loss and gain of electrons equal. In the half-reaction method, you determine the oxidation numbers and write two half-reactions. You are making the total loss of electrons equal to the total gain of electrons. Then you multiply the atoms that have changed by small whole numbers. In the oxidation number method, you determine the oxidation numbers of all atoms. Then you balance by making the electron loss equal the electron gain. The only sure-fire way to balance a redox equation is to recognize the oxidation part and the reduction part. They are just different ways of keeping track of the electrons transferred during the reaction. There's no real difference between the oxidation number method and the half-reaction method. Now try to balance the equations in the link below (answers included). Place these numbers as coefficients in front of the formulas containing those atoms.ĢHNO₃ + 3H₃AsO₃(aq) → 2NO(g) + 3H₃AsO₄(aq) + H₂O(l)īalance all remaining atoms other than H and O. This gives us total changes of -6 and +6. We need 2 atoms of N for every 3 atoms of As. Make the total increase in oxidation number equal to the total decrease in oxidation number. Right hand side: N = +2 O = -2 H = +1 As = +5ĭetermine the change in oxidation number for each atom that changes. Left hand side: H= +1 N= +5 O = -2 As = +3 Identify the oxidation number of every atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
